Green Ammonia Production
To produce Green Ammonia you need three basic ingredients: air, water, and an abundant source of renewable electricity. Not to forget investment capital, industrial technology, and a means to get the product to market ! Huge areas of the world are suitable for making Green Ammonia, however most production will be concentrated in places where renewable energy cost is lowest, and access to markets is most favourable.

Image: schematic diagram of the entire Green ammonia production cycle.
First, make your Hydrogen …
All industrial scale ammonia production in the world today begins by making hydrogen (typically using natural gas as the hydrogen and energy source), then chemically combining it with nitrogen (separated from the air).
This last step is done using the Haber (or Haber Bosch) process, in which the two gases are subjected to high temperature and pressure in the presence of a catalyst.
Green ammonia is made using the same two steps, but using water as the hydrogen source (via electrolysis), and with all the required energy generated renewably. If the best available water source is the sea, as may often be the case in hot, sunny countries rich in solar power, desalination will first be required.
There are also alternative approaches to Green ammonia synthesis which avoid the manufacture of hydrogen as an intermediate stage. These are discussed at the end of this page.
Desalination of Seawater
Where fresh water is scarce, sea water can be made suitable for electrolysis by desalination. Hydrogen composes 2/18 by mass of water, and 3/17 by mass of ammonia, so it takes
18/2 x 3/17 = 1.6 tonnes of water per tonne of ammonia.
With Reverse Osmosis, it takes 3.5 - 4.5 kWh of electricity (average 4) to make a tonne of desalinated water. So desalination represents an energy cost of 8 kWh per tonne of Green Ammonia - negligible compared to other inputs.
Brine disposal
Care must also be taken with the disposal of brine wastes. Typically every tonne of fresh water produced by desalination creates a tonne of brine, at double the marine salt concentration. These brines are often returned to the sea. However they’re toxic to sea life, and being heavy, sink to the bottom of the water.
In 2020 some 1.6 billion cubic metres of such brines were generated from desalination mainly for fresh water supply. The large scale production of hydrogen / ammonia in hot, dry tropical regions could create a several fold increase in that figure.
Good practice for brine disposal is to dilute it in seawater and pump it by submarine pipeline to a zone of naturally high water circulation to ensure dispersal.
It may also be possible, and economic, to use desalination brines as a source of valuable metals and other elements.

Image: Desalination: producing fresh water from the sea, and returning salty brines.

Photo: The Azzazia desalination plant in Saudi Arabia, Credit: Waleed Alzuhair via Flickr, CC BY-NC-SA 2.0.
Hydrogen Electrolysis
Electrolysis splits the water into hydrogen and oxygen (which may be sold, or simply released back to the atmosphere).
To produce 1 tonne of hydrogen, with an energy value of 120 GJ (33.3 MWh) / takes approximately 52.5 MWh of electricity using currently available ‘alkaline PEM electrolysis’ technology. This represents a 63.5% conversion efficiency.
One tonne of hydrogen can make 5.7 tonnes of ammonia, so this represents an energy input of 9.7 MWh per tonne of Green ammonia.
Clearly, the 63.5% efficiency figure is one that has much scope for improvement, and if substantially raised would transform both the energy and performance and financial attractiveness of the Green hydrogen / ammonia energy system.
New Technologies
A number of new electrolysis technologies are in the offing, and efficiencies are expected to rise to some 75% for PEM systems and 80% for Solid Oxide electrolysers.
But most promising is the ‘bubble-free’ approach taken by Hysata, an Australian company which promises an astonishing “95% efficiency” for its forthcoming product, taking the energy of electrolysis down to just 41.5 MWh / tonne of hydrogen. This is presumably based on the Higher Heating Value (HHV) of hydrogen (39.39 NWh/kg). Using the Lower Heating Value we obtain a still remarkable and transformative efficiency of 80%.
As the company points out, “electricity makes up most of the cost of green hydrogen. The more energy-efficient the electrolyser, the less electricity is wasted, and the lower the cost of green hydrogen. Less waste heat also translates to a simpler balance of plant with less cooling and liquid handling required.
“Hysata’s transformative electrolysis design is underpinned by two key innovations: an ultra-low resistance separator, and bubble-free operation. Together, these factors collapse the resistance of the cell and make our cell highly efficient.”
Standard alkaline PEM electrolysis works best with a firm, steady power supply, as provided, for example, by a grid connection or a dedicated hydroelectric plant. However we also need electrolysers whose output is easily modulated according to the variable power output by wind and solar generators.
These are now available, for example from the Japanese hi-tech materials company AGC, which in 2024 announced a new mass production facility for its FORBLUE S-SERIES fluorinated ion-exchange membrane.

Figure 2: Electrolysis of water to hydrogen and oxygen.

Image: Schematic representation of a typical alkaline electrolyser. Credit: Kavin Teenakul / CC BY-SA 4.0.
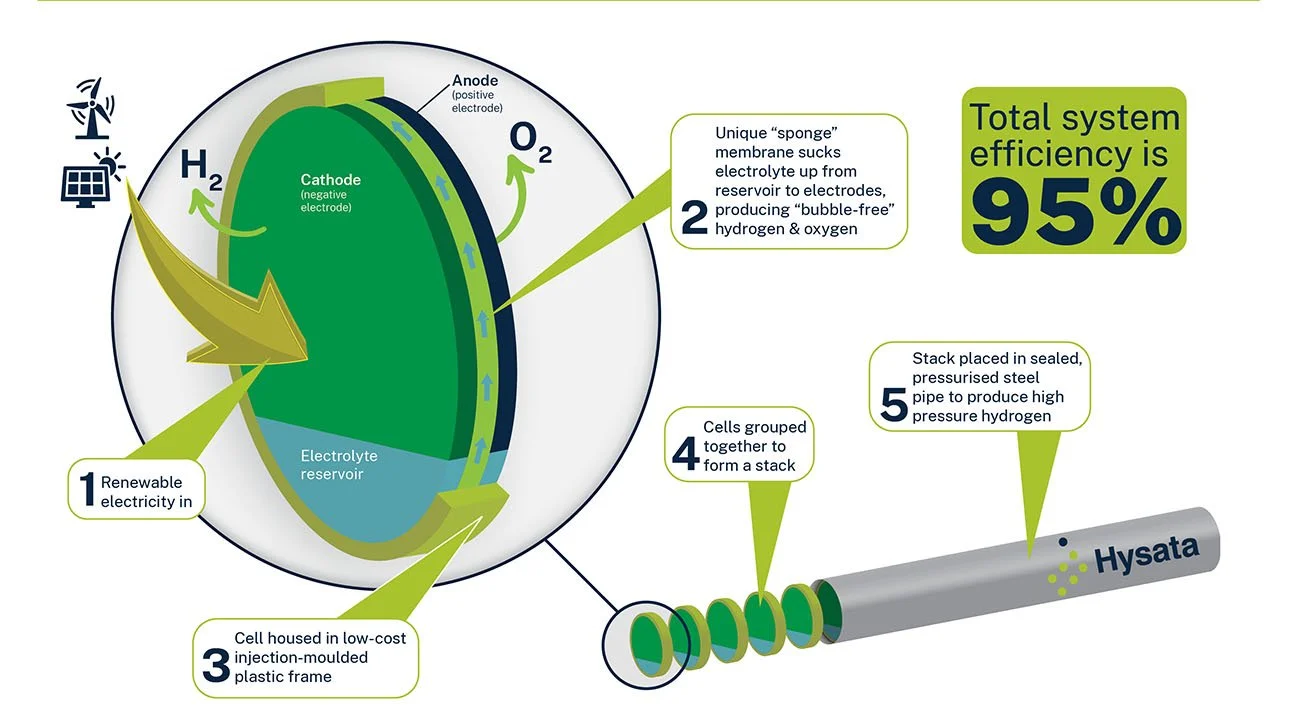
Image: Schematic depiction of Hysata’s ‘bubble-free’ hydrogen electrolysis technology, which retains 80% of input electrical energy in the thermal value of the hydrogen produced - up from the currently typical 63.5%. Credit: Hysata.
Nitrogen Separation from Air
The nitrogen needed for the ammonia generation is readily available from the ambient air, which is 78% nitrogen.
The nitrogen is most efficiently extracted from air using pressure swing adsorption (PSA) technology, which uses ‘molecular sieves’ (often zeolites or activated carbon) to adsorb the nitrogen from air under high pressure, then release it in a low pressure phase.
PSA uses 0.31 to 0.63 kWh / kg of nitrogen gas. The electrical energy needed for traditional cryogenic air separation is 2.56 kWh / kg of liquid nitrogen.
To make one tonne of ammonia needs 14/17 tonnes = 824 kg of nitrogen. Using PSA we get an energy use of 255 - 519 kWh / tonne N2. Cryogenic separation is much higher at 2.1 MWh / tonne.
To produce enough nitrogen for one tonne of Green ammonia we adopt the lowest possible figure reflecting the best and newest technology, hence 255 kWh.

Photo: A small pressure swing adsorption (PSA) nitrogen generator. Credit: Nicky Xiong via Wikimedia Commons CC-BT-SA 2.0.
Haber-Bosch Process
The Haber-Bosch (or Haber) process reacts nitrogen with hydrogen at high temperature (300-500C) and pressure (80-300 atmospheres) in the presence of a metallic catalyst. To make a tonne of ammonia requires 176kg of hydrogen and 824kg of nitrogen.
Although the ammonia forming reaction is exothermic, the process consumes significant energy: both to reach the required temperature; and to separate the ammonia from the hydrogen and nitrogen reagents in successive cycles.
The electricity consumption of the Haber Bosch loop including ammonia separation is currently 0.6-0.7 MWh/t NH3 (Laura Torrente Murciano, pers.comm). Let’s call it 0.65 MWh per tonne. Improvements will be warmly welcomed in the Green ammonia industry!

Photo: The Haber process ammonia synthesis plant at Billingham, UK, which has been fuelled by natural gas since the 1970s. Credit: Mick Garrett / Wikimedia Commons CC BY-SA 2.0.

Image: A flow diagram for the synthesis of ammonia using the Haber process, using methane as the fuel and hydrogen source. For making Green ammonia from hydrogen and nitrogen, the process begins at the Compressor in the centre of the image. Credit: Wikimedia Commons CC-BY-SA 4.0.
Energy Summary
Adding up the energy needed to produce a tonne of Green ammonia from the figures above we get:
(0.008 + 9.7 + 0.255 + 0.65) MWh ~= 10.6 MWh.
So 91.5% of the total energy cost of making Green ammonia is taken up in the initial production of hydrogen, while 6.1% is taken up by ammonia synthesis and 2.3% arises from the nitrogen separation.
To achieve significant reductions in the required energy inputs the first priority is to raise the efficiency of electrolysis, and of the Haber-Bosch process. Or, alternatively, to develop an entirely new and better way of doing it.

Image: With more than half of the energy of Green ammonia production going in electrolysis, efficiency improvements as offered by this bubble-free alkaline capillary-fed electrolysis (CFE) cell are highly sought after. Credit: University of Wollongong, CC BY 4.0.
Alternative routes to Nitrate Fixation
There are alternative approaches to Green ammonia synthesis which avoid the manufacture of hydrogen as an intermediate stage. None of these are current commercially viable, however some might become so with further research and investment.
Historically, ‘fixed’ (non-gaseous) nitrogen has been scarce and highly valued as a plant nutrient. The greatest primary sources of fixed nitrogen were: the action of soil and marine bacteria; and the action of lightning, which provided the energy to rupture the powerful triple bond of the N2 molecule and allow it to bond instead with oxygen, forming NOx species.
Thus the Birkeland-Eyde process (see image), first developed in 1903, works by the direct fixation of NOx species from air in the plasma phase through high voltage electrical discharges. The NOx is then fully oxidised to nitrate. This approach fell into disuse following the development of the far more efficient Haber process.
However modern variants of Birkeland-Eyde (for example) and other novel processes, have the potential to make small scale ‘on farm’ Green nitrate (and ammonia) synthesis more viable, and could ultimately reduce costs for larger scale production.

Image: Different nitrogen fixation processes: (a) Birkeland & Eyde process for nitrate synthesis (b) Haber process for ammonia synthesis. Credit: Ashadul Adalder et al, Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International.

Photo: to you and me it’s just mist, but for scientists it’s a cornucopia of possibilities for chemical synthesis that transcends the boundaries of conventional thermodynamics. Credit: Serchfdlopez via Weather Wiki.

Photo: the Stanford Chemistry Department, home to Zarelab where the novel ammonia synthesis process driven by microdroplets of water was discovered. Credit: Jun Seita via Flickr CC-BY 2.0.
Stanford’s Magic Microdroplets
Chemists at Stanford University in California recently discovered that ammonia can be synthesised using the mysterious power of highly reactive microdroplets of water.
In their experiment, Stanford chemists Xiaowei Song, Chanbasha Basheer and Richard Zare created a spray of droplets with a median diameter of ten microns, expelled through fine capillary tubes at a mesh coated with Nafion (as proton donor) and ferrous ferric oxide (Fe3O4) as catalyst.
Mass spectrometer analysis revealed the presence of both ammonia and hydrazine (H2NNH2), apparently an intermediate stage of the ammonia synthesis.
According to the authors’ PNAS article, “Mounting evidence shows that water microdroplets can not only facilitate the one-electron (e-) reduction due to its unique air-water interface redox environment but also capture reactive carbocation intermediates promoted and stabilized by its interfacial hydronium ions (H3O+).
“Therefore, we suggest that the abundance of H3O+ and e- at the microdroplet interface also plays a role in the hydrogenation of N2 molecules bound to the Fe3O4 surface when in contact with water microdroplets, forming H2NNH2 and ultimately NH3.”
“It is worth noting”, they add, “that the ammonia production process reaches an optimum value when neither external potential nor extra heat is applied to the catalyst mesh or to the spray source, avoiding electric or thermal power consumption.”
The ammonia yield is small, estimated at 33 nanomoles per second per square centimetre of catalytic mesh. At this rate 100 square metres of mesh would need to be sprayed with water microdroplets for six hours to make one kilo of ammonia.
Much work will be needed to increase the rate of ammonia production before the process has practical use. But for now the extraordinary thing is that ammonia is made at all. And as Zare told the Stanford Report, it took five years for the Haber process to reach maturity.
“If it really works, it’s huge,” said Zare. “It would cut the cost of food, and it would help us in regard to climate change. It’s huge if it really can be done.”
References
S. M. Saba, M. Müller, M. Robinius, and D. Stolten. The investment costs of electrolysis – a comparison of cost studies from the past 30 years. International Journal of Hydrogen Energy, 43(3):1209–1223, 2018.
C. Smith, A. K. Hill, and L. Torrente-Murciano. Current and future role of Haber–Bosch ammonia in a carbon-free energy landscape. Energy Environ. Sci., 13:331–344, 2020.
